|
For example, one author asserts that $\pi = 17 – 8 \sqrt.$$ These formulas are entirely satisfactory to calculate the semiperimeters and areas of inscribed and circumscribed circles, provided one has a calculator or computer program to evaluate tangents and sines. 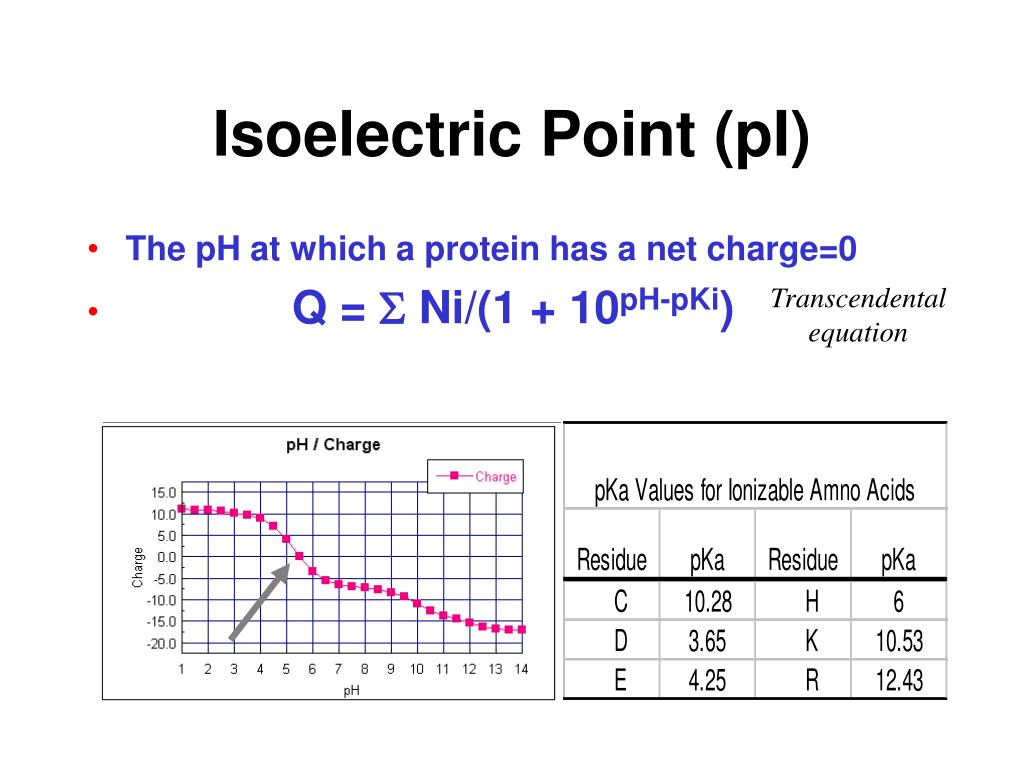
One motivation for this article is to respond some recent writers who reject basic mathematical theory and the accepted value of $\pi$, claiming instead that they have found $\pi$ to be a different value. We will also discuss zwitterions, or the forms of amino acids that dominate at the isoelectric point. You will learn how to calculate the isoelectric point, and the effects of pH on the amino acids overall charge. For a step-by-step presentation of Archimedes’ actual computation, see this article by Chuck Lindsey. The isoelectric point of an amino acid is the pH at which the amino acid has a neutral charge. In this article, we present Archimedes’ ingenious method to calculate the perimeter and area of a circle, while taking advantage of a much more facile system of notation (algebra), a much more facile system of calculation (decimal arithmetic and computer technology), and a much better-developed framework for rigorous mathematical proof. For additional details, see the Wikipedia article. Indeed, with this method Archimedes anticipated, by nearly 2000 years, the modern development of calculus that began in the 17th century with Leibniz and Newton. But his most far-reaching discovery was the “method of exhaustion,” which he used to deduce the area of a circle, the surface area and volume of a sphere and the area under a parabola. He was a pioneer of applied mathematics, for instance with his discovery of the principle of buoyancy, and a master of engineering designs, for instance with his “screw” to raise water from one level to another. In general, the pI values for native proteins are affected by the three-dimensional structure of the proteins, which causes greater differences between calculated and experimental pI values than in the case of polypeptides for which pI values are determined in the presence of urea.Archimedes is widely regarded as the greatest mathematician of antiquity. The pI of the native Glut1 was lower, 8.0 /- 0.1, at 22 degrees C. The calculated pI for the human red cell glucose transporter (Glut1) with one sialic acid residue was decreased from 8.8 to 8.5 by introducing pKa value spreading and became consistent with the experimental pI value of 8.4 /- 0.05 at 15 degrees C determined in the presence of 6 M urea. The calculated pI values showed reasonably good agreement with experimental ones for most of 16 native proteins over a wide pH range (3.4-11) when charge contributions of heme groups, sialic acid residues, etc., were taken into account. Each particular type of ionizable group was assumed to have pKa values distributed around the chosen value, thereby simulating the situation in proteins and polypeptides. 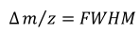
A set of pKa values was chosen for amino acid residues with ionizable side chains. 
Amino acid composition, pKa values for amino acid side chains and for the N- and C-terminal groups, and the presence of other charged groups were taken into account. For estimating pI values the net charge of several proteins was calculated versus pH by use of the Henderson-Hasselbalch equation. The isoelectric points (pI) of native proteins are important in several separation techniques.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
